Lactic acid: good or bad?
Dr Sarah Powell and Robert Stockdale examine the current understanding of lactic acid to assess its role in the body

EXAM LINKS
AQA Energy systems, energy transfer in the body and energy transfer during short-duration/high-intensity exercise
Edexcel Energy systems, fatigue and recovery
OCR Energy for exercise, energy systems and ATP resynthesis
Lactic acid is that pesky substance that causes burning, pain, muscular fatigue and poor performance. Or does it? Lactic acid is generally the accepted rogue of training and performance, but there is much more to this than meets the eye.
Historically, the notion that lactic acid causes muscle fatigue was due to a correlation between lactate accumulation in the blood and the onset of fatigue symptoms during exercise. However, correlation does not mean causation.
In 1907, W. M. Fletcher and F. Gowland Hopkins stimulated frog muscles to contract. They observed fatigue in line with blood lactate accumulation, and the disappearance of lactate when the fatigued muscles were placed into an oxygen-rich environment. In 1922, the Nobel prize in physiology or medicine was divided equally between Archibald Vivian Hill for discovering heat production in skeletal muscle, and Otto Fritz Meyerhof for discovering a fixed relationship between oxygen consumption and the metabolism of lactate in the muscle.
Both studies attributed lactic acid production to a lack of oxygen and the basis for the idea that lactic acid causes muscular fatigue under anaerobic conditions was set. It was theorised that the accumulation of lactic acid lowered blood and muscle pH through acidosis (high acidity in the blood or muscle tissue), which reduced enzyme function. These acidic conditions reduce the function of calcium ions and interfere with the mechanism of muscle contraction.

A turning point in the debate came in 2004 when Robert Robergs proposed a lack of biochemical support for lactic acid causing acidosis and in turn, fatigue, at any given intensity of exercise. This instigated the debate, continuing today, as to whether lactic acid really does cause fatigue.
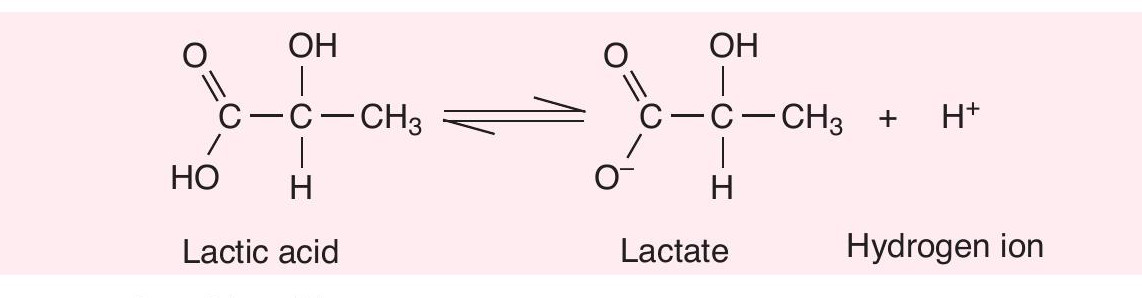
Lactic acid separates into lactate and a hydrogen ion (H+ , Figure 1.1). Currently, the most widely accepted cause of fatigue during exercise is the accumulation of hydrogen ions rather than the accumulation of lactic acid, as previously suggested.
Box 1 Lactic acid v lactate
Lactic acid is commonly used as an alternative term to lactate. Lactic acid is the protonated (with an added proton) form of lactate, meaning the two compounds are clearly distinguishable by the presence of an additional hydrogen ion (H+) in the chemical structure of lactic acid.
Formation
When we start to exercise at a high intensity the demand for adenosine triphosphate (ATP) from the muscles dramatically increases. This demand is not matched by the supply of oxygen as there simply is not time. Until oxygen use is plentiful a performer must rely on anaerobic ATP production.
Anaerobic glycolysis (Figure 1) occurs in the sarcoplasm and involves the breakdown of glucose in the absence of oxygen. The regulatory enzyme is phosphofructokinase (PFK) and there is a net gain of enough energy to resynthesise two moles of ATP. The product of glycolysis is pyruvate, which — without the presence of oxygen — is reduced to form lactate (a reaction catalysed by the enzyme lactate dehydrogenase, LDH). The important but often overlooked products of this process are hydrogen ions.
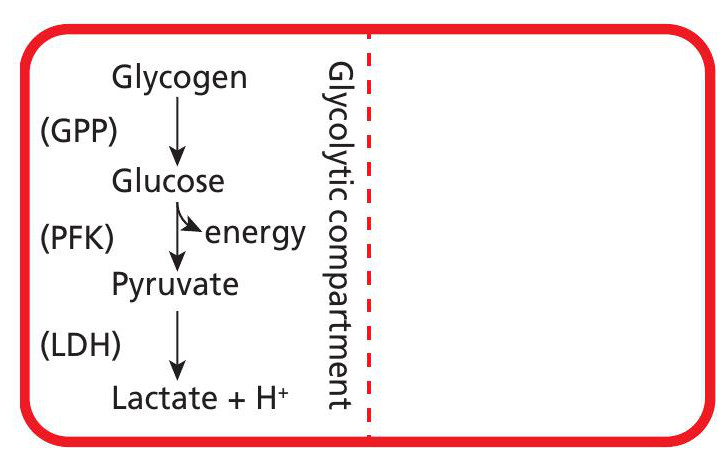
The production of lactate and hydrogen ions is suggested to occur in a glycolytic compartment of the muscle cell close to the myofibrils (the contractile units of a muscle cell) to directly benefit from stores of glycogen. Once produced, lactate is transferred out of the muscle cell and directly into the venous bloodstream.
Performance impact
The onset of blood lactate accumulation (OBLA) is characterised by a blood lactate concentration greater than 4 millimoles per litre (mmol/l). Reaching OBLA decreases exercise performance.
In untrained individuals OBLA may occur at around 65% of VO2 max, whereas in highly trained individuals this may occur at around 85% of VO2 max (Figure 2). For both untrained and trained performers blood lactate will remain stable at around 1–2 mmol/l if the exercise intensity is low to moderate. As intensity rises lactate production exceeds removal and begins to accumulate (known as the lactate threshold). After around 4mmol/l blood lactate rises exponentially — the lactate turn point — and will have a significant and fast impact on performance.
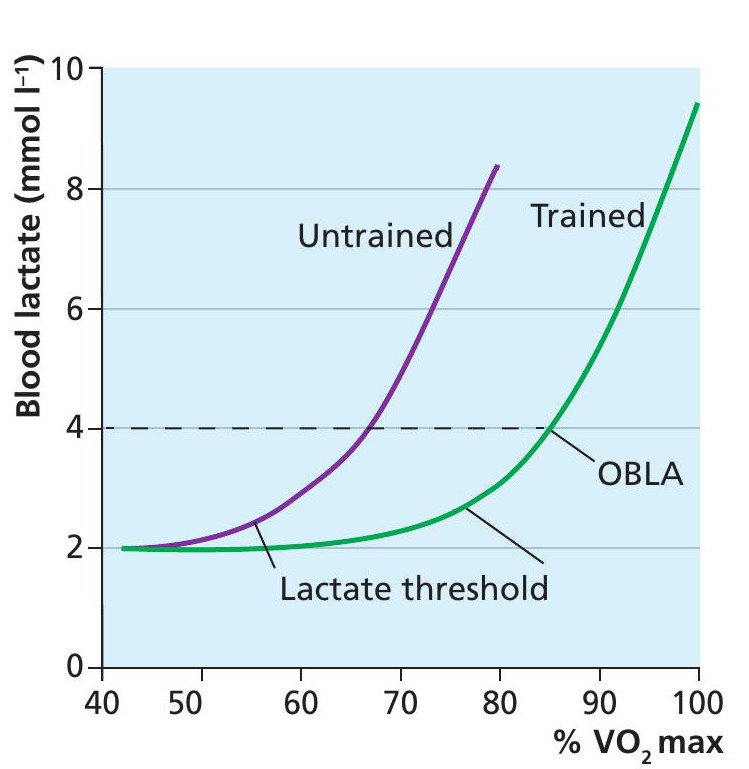
A well-trained performer’s blood lactate curve shifts to the right. They reach OBLA at a higher percentage of their VO2 max compared to untrained individuals. This means they can train and perform at higher intensities or for longer before their performance is affected.

Box 2 Did you know?
Research on what it takes to run a sub-2- hour marathon states that, for a 59 kg runner a VO2 max of 67 ml/kg/min would be required, with the exponential rise in blood lactate accumulation only occurring at 92% VO2 max. Research Nike’s ‘Breaking2 project’, led by Professor Andrew Jones, to find out more.
How does training help limit the onset of OBLA?
1 Increased mitochondrial density and enzyme activity lead to an increased oxidative capacity of skeletal muscle in trained performers. They can take in and use more oxygen, which reduces the reliance on anaerobic metabolism and lactate accumulation at any given running velocity.
2 Increased skeletal muscle capillary density in trained performers means metabolites — including lactate and hydrogen ions — are removed more effectively, which reduces the acid environment.
3 Increased activation of lactate transporters (such as MCT1) in trained performers means blood lactate is more readily recycled reducing the acid environment.
These adaptations, as well as an efficient cardio-respiratory system, contribute to trained individuals having an increased ability to supply, transport and use oxygen, and to tolerate and remove lactic acid.

Good or bad?
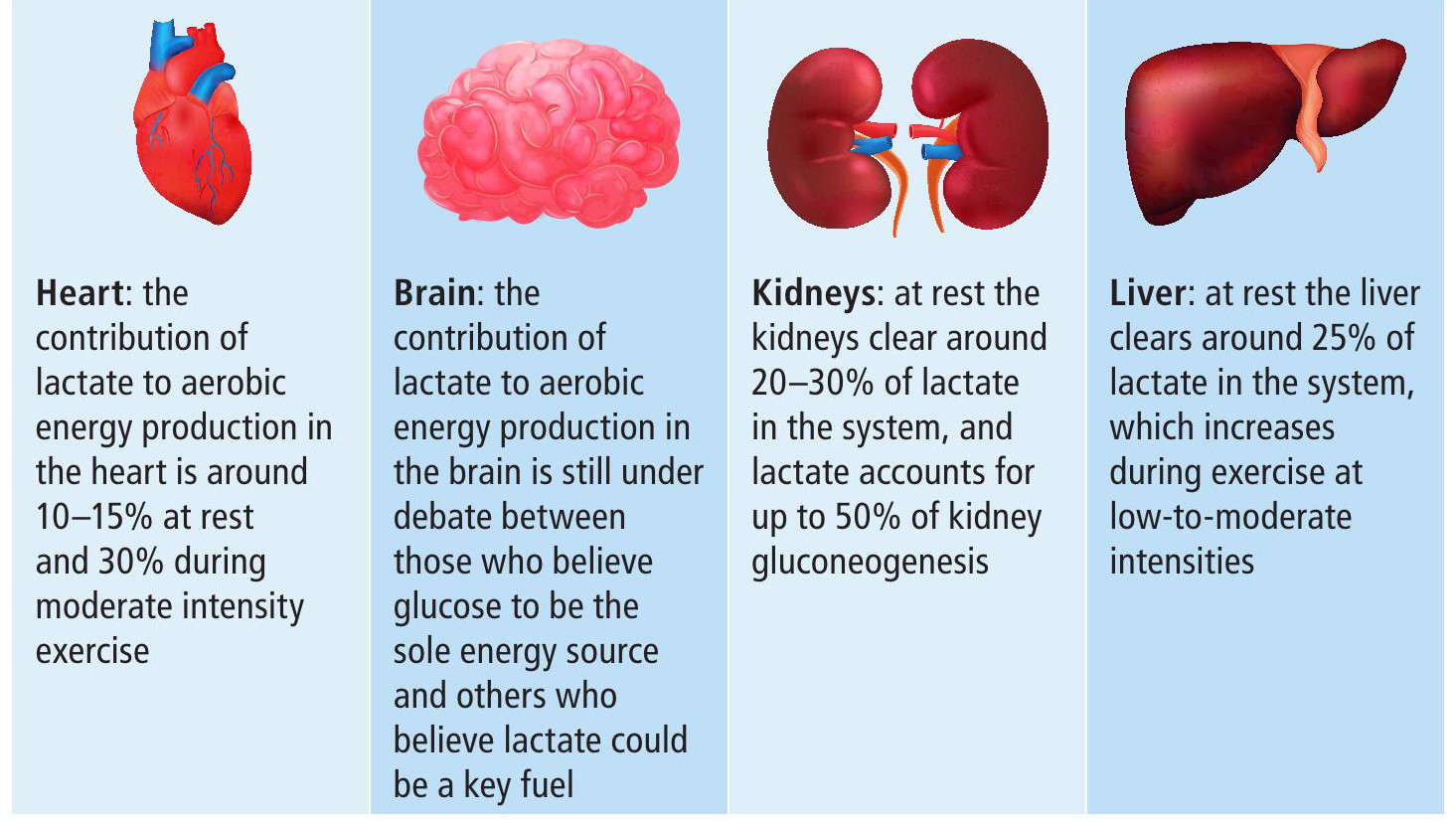
Lactic acid may not be the villain, but can it ever be considered a hero? The muscle cell is well tuned to lactate and has an exceptional ability to consume it as well as produce it. Lactate can enter an oxidative compartment close to the mitochondria, where it is converted back into pyruvate and used as a fuel for the Krebs cycle and aerobic energy production (Figure 3).
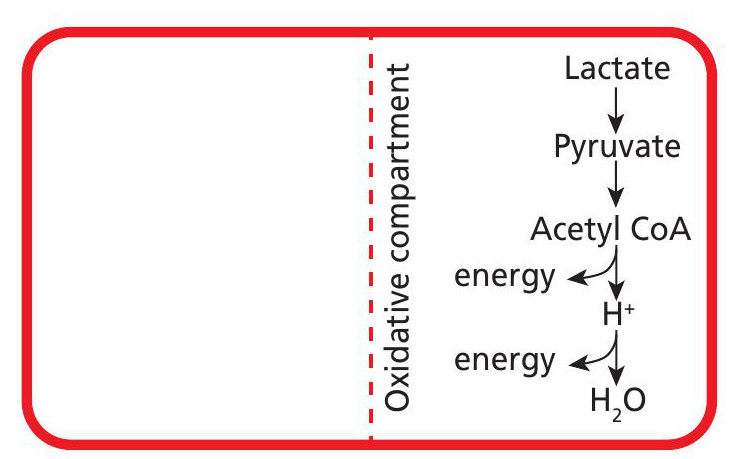
Therefore, lactate is highly beneficial for energy turnover within the muscle cell without wasting the valuable resources of glucose and glycogen. Equally, lactate can be drawn from the circulation and directly oxidised without first being converted into glucose (gluconeogenesis) by the liver or kidneys — a far more direct approach.
Benefits
1 Hydrogen ions released are consumed during the LDH reaction acting as a buffer against rises in acidity and, in turn, fatigue.
2 Lactate produced from the LDH reaction is removed from the cell via transporters (such as MCT4), which have been found to help remove hydrogen ions from skeletal muscle.
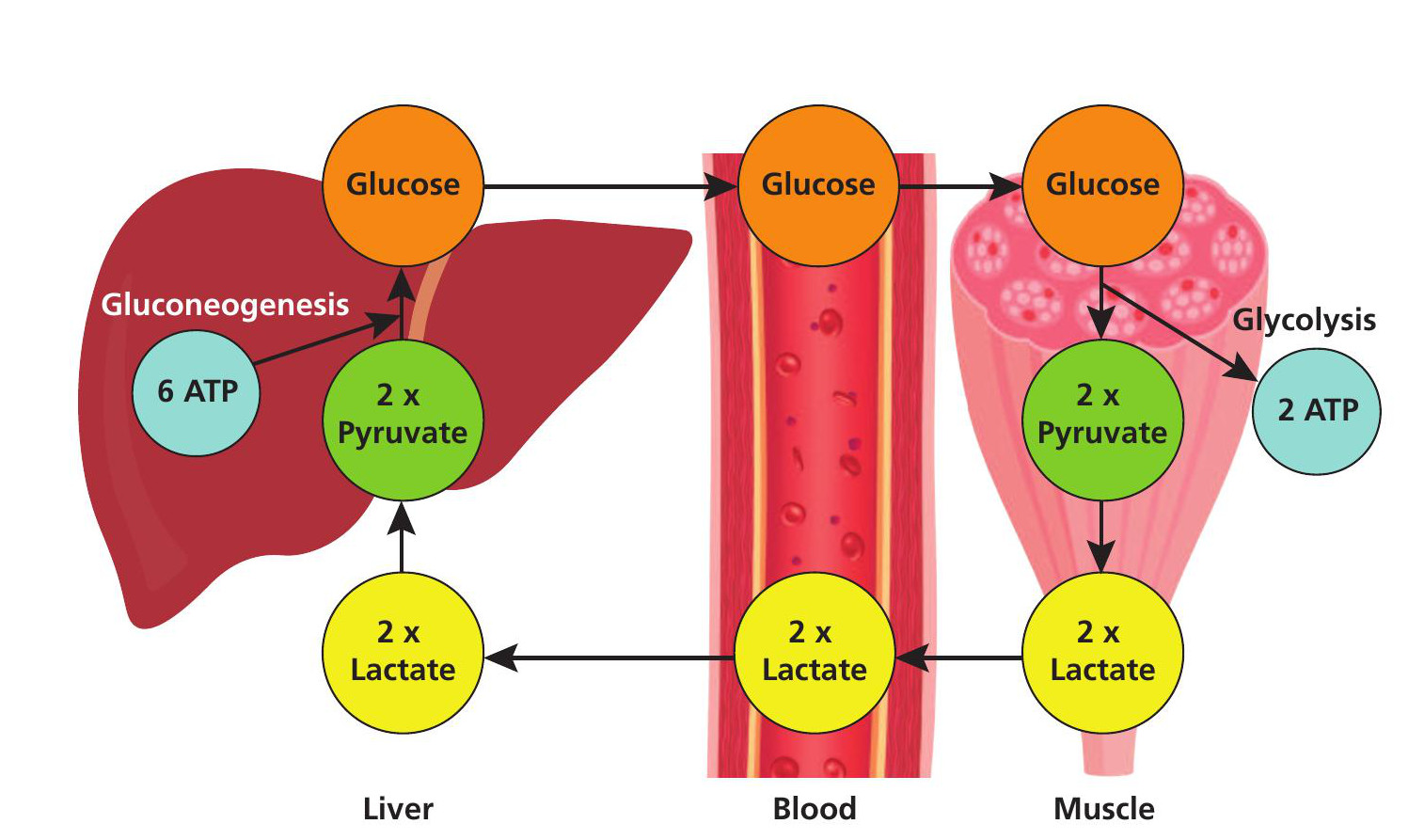
3 Lactate removed from skeletal muscle is circulated away to be taken up and used as a substrate for metabolism in other tissues, for example the Cori cycle in the liver removes around 25% of lactate via gluconeogenesis (Figure 4). This provides another fuel for glycolysis and ATP resynthesis.
Ultimately, the research is ongoing and there are more discoveries to come. One thing we do know is that lactic acid itself does not cause fatigue.
KEY TERMS
Anaerobic glycolysis The partial breakdown of glucose to pyruvate in the sarcoplasm.
Buffering The ability of hydrogen ions (buffers) to neutralise the effects of blood lactate.
Catalyst Substance that increases the rate of, or facilitates, a chemical reaction.
Cori cycle A metabolic pathway where lactate produced during anaerobic glycolysis is transported to the liver and converted back into glucose.
Electron transfer chain The third stage of the aerobic system producing energy to resynthesise 34 moles of ATP in the mitochondrial cristae.
Gluconeogenesis The formation of glucose from a substrate, such as pyruvate.
Glyconeogenesis The formation of glycogen from a substrate, such as pyruvate.
Krebs cycle The second stage of the aerobic system producing energy to resynthesise 2 moles of ATP in the mitochondrial matrix.
Mole A scientific unit of measurement.
OBLA Onset of blood lactate accumulation. The point where a dramatic rise in blood lactate levels occur.
Sarcoplasm Cytoplasm of a muscle fibre.
Dr Sarah Powell is a teaching fellow at Lancaster University Medical School and author of a range of A-level PE textbooks and resources.
Robert Stockdale is a postgraduate student studying at Lancaster University for a MSc by research in training and match play forces and velocity in elite male footballers.